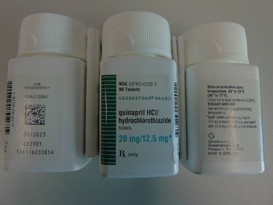
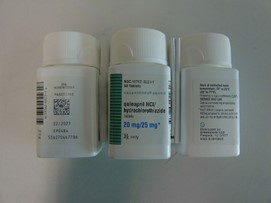
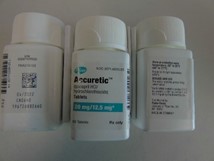
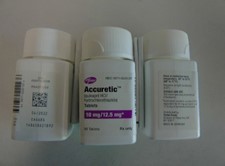
Pfizer will recall six lots of Accuretic tablets, one lot of quinapril and hydrochlorothiazide tablets and four lots of quinapril HCl/ hydrochlorothiazide tablets.
The company is voluntarily recalling Accuretic (quinapril HCl/hydrochlorothiazide) tablets distributed by Pfizer as well as two authorized generics distributed by Greenstone (quinapril and hydrochlorothiazide and quinapril HCl/ hydrochlorothiazide) to the patient (consumer/user) level due to the presence of a nitrosamine, N-nitroso-quinapril, above the Acceptable Daily Intake (ADI) level.
Nitrosamines are common in water and foods, including cured and grilled meats, dairy products, and vegetables. Everyone is exposed to some level of nitrosamines. These impurities may increase the risk of cancer if people are exposed to them above acceptable levels over long periods.
These products are indicated for the treatment of hypertension. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. ii The products have a safety profile that has been established over 20 years of marketing authorization and through a robust clinical program. To date, Pfizer is not aware of reports of adverse events that have been assessed to be related to this recall. Pfizer believes the benefit/risk profile of the products remains positive based on currently available data. Although long-term ingestion of N-nitroso-quinapril may be associated with potential increased cancer risk in humans, there is no immediate risk to patients taking this medication. Patients currently taking the products should consult with their doctor about alternative treatment options.
Pfizer presented photos of the products. More information on the company website.
Accuretic™ (quinapril HCl/hydrochlorothiazide) tablets

Quinapril and hydrochlorothiazide tablets

quinapril HCl/hydrochlorothiazide tablets